Tag: cellular
iOS 16.4 Adds Voice Isolation for Cellular Phone Calls
Apple says that Voice Isolation will prioritize your voice and block out the ambient noise around you, making for clearer phone calls where you can better hear the person you’re chatting with and vice versa.
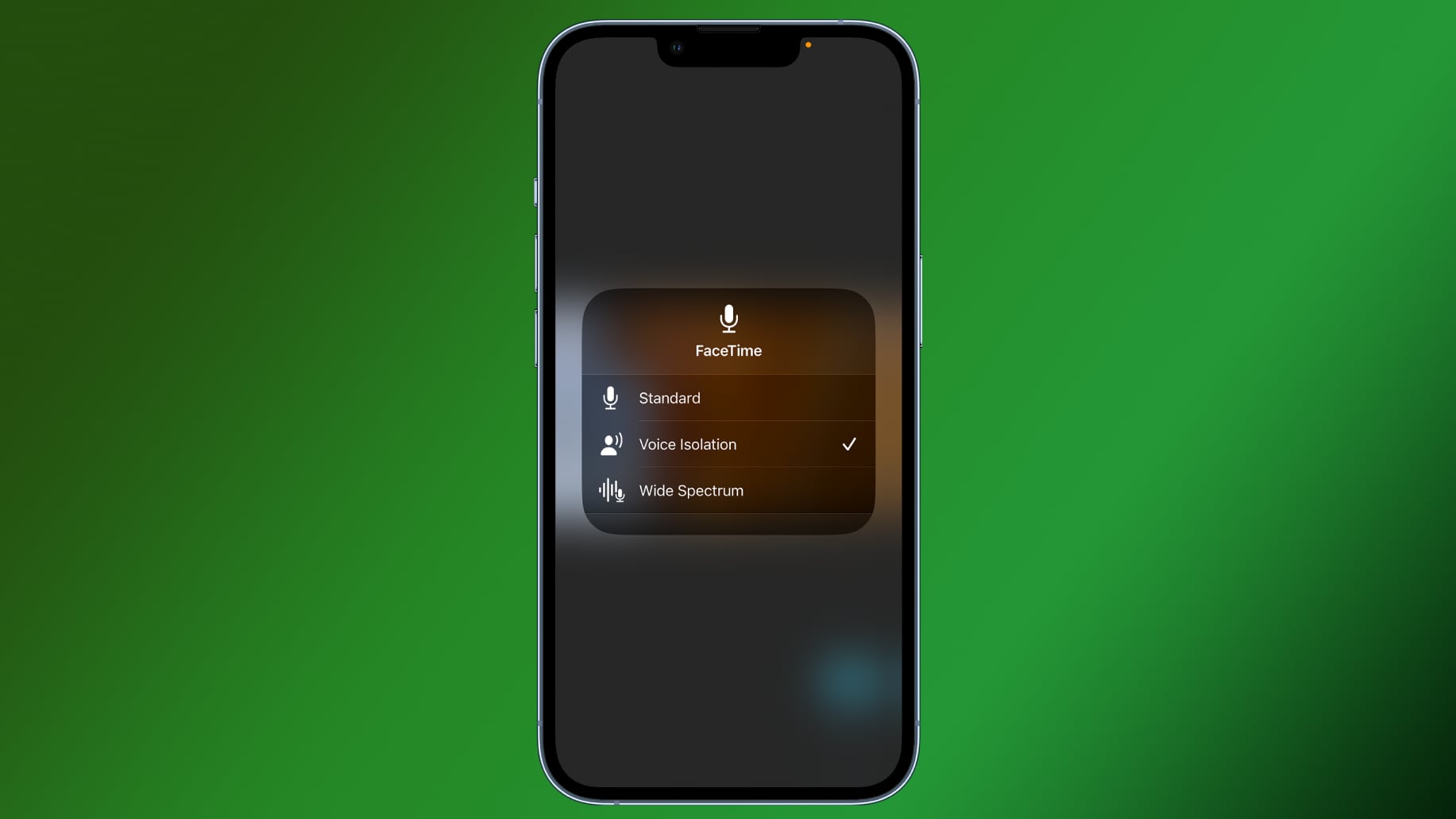
Voice Isolation has been available for VoIP calls that use FaceTime, WhatsApp, and other apps on devices running iOS 15 or macOS Monterey or later, and it is designed to significantly improve microphone quality when on calls by cutting down on background noise. Until now, it was not available for cellular conversations, with Apple offering no way to improve call quality.
On a FaceTime call, Voice Isolation can be enabled by activating control center, tapping on the Mic Mode option, and choosing the Voice Isolation option, so the feature may be able to be activated in a similar way for voice calls.
Other iOS 16.4 features include new emojis, Push Notifications for web apps, bug fixes for HomeKit, Crash Detection optimizations, and more.
This article, “iOS 16.4 Adds Voice Isolation for Cellular Phone Calls” first appeared on MacRumors.com
Discuss this article in our forums
Google Fi Reportedly Drops US Cellular, Leaving T-Mobile As Last Network – CNET
Apple is reportedly making an all-in-one cellular, Wi-Fi, and Bluetooth chip

Apple is working on a new in-house chip that would power cellular, Wi-Fi, and Bluetooth functionality on its devices, according to a report from Bloomberg. The company is also developing its own chip that would replace the Wi-Fi and Bluetooth chip it currently uses from Broadcom, Bloomberg says, which it wants to begin using in devices in 2025.
Bloomberg also shared some new information about Apple’s efforts to develop its own cellular modems to replace Qualcomm’s. While Qualcomm recently said during its Q4 2022 earnings that it expects to have the “vast majority” of 5G modems for 2023 iPhones, Bloomberg says Apple will use its own modems “by the end of 2024 or early 2025.” Apple will apparently start by using its custom modem in one…
Daily Authority: 📡 From cellular to satellite
Apple and Ericsson Reach Licensing Agreement to Settle Dispute Over Cellular Patents

The dispute dates back to early 2015 when the two companies sued each other over dozens of Ericsson patents related to cellular technology used in Apple products such as the iPhone and iPad.
The companies reached a seven-year patent licensing agreement in late 2015 that appeared to put an end to the dispute, but it was revived in late 2021 and early 2022 as the 2015 agreement neared its end and the companies were unable to agree on terms for extending the agreement and incorporating additional patents related to newer 5G technology.
With today’s announcement, Ericsson and Apple have entered into a new multi-year agreement for cross-licensing cellular-related patents and additional patent rights.
Christina Petersson, Chief Intellectual Property Officer at Ericsson says: “We are pleased to settle the litigations with Apple with this agreement, which is of strategic importance to our 5G licensing program. This will allow both companies to continue to focus on bringing the best technology to the global market.”
In addition to the patent licensing, the agreement also includes commitments from both companies to strengthen their existing collaborations, “including in technology, interoperability and standards development.”
This article, “Apple and Ericsson Reach Licensing Agreement to Settle Dispute Over Cellular Patents” first appeared on MacRumors.com
Discuss this article in our forums
Apple and Ericsson call truce in years-long fight over cellular patents
Apple is ending another battle over wireless patents. The iPhone maker and Ericsson have struck a licensing deal that settles all the legal disputes between the two companies, including civil lawsuits and a US International Trade Commission complaint. While the exact terms remain under wraps, the multi-year pact includes cross-licensing for “standard-essential” cellular technology as well as other patent rights.
The tech giants have a long history of fighting over cell tech. Apple sued Ericsson in 2015 to get more favorable terms for LTE patents, but Ericsson responded with a lawsuit of its own claiming that the iPhone and iPad infringed on its patented ideas. The two achieved peace with a seven-year agreement. As that arrangement neared its renewal time, however, the animosity returned. Ericsson sued in October 2021 over Apple’s attempts to shrink royalty rates, while Apple countersued in December that year over allegations Ericsson was using unfair pressure tactics for the renewal. Ericsson filed another lawsuit this January over 5G licenses.
We’ve asked Apple for comment. In announcing the deal, Ericsson’s IP chief Christina Petersson said the ceasefire would let the two companies “focus on bringing the best technology” to the world. Ericsson is one of the world’s largest wireless patent holders, and said the Apple agreement would help boost its licensing revenue for the fourth quarter to the equivalent of $532 million or more.
The timing may be significant. Apple is reportedly developing 5G iPhone modems to replace Qualcomm’s chips, having bought most of Intel’s modem business and even launching not-so-subtle recruitment efforts in Qualcomm’s backyard. The Ericsson truce may help clear the path for those modems by reducing the chances of legal dust-ups over whatever Apple builds. And time might be in short supply — rumors have circulated that Apple could use its own components as soon as 2023.
The best US Cellular holiday deals: Get any phone for free plus $100
Deals: Amazon Has AirPods Pro 2 for $234 ($15 Off) and Cellular Apple Watch Series 8 for $389 ($110 Off)
AirPods Pro 2
Amazon today has the AirPods Pro 2 for $234.00, down from $249.00. This is one of the first times we’ve seen a discount on the second-generation AirPods Pro since Prime Early Access in October.
 Note: MacRumors is an affiliate partner with some of these vendors. When you click a link and make a purchase, we may receive a small payment, which helps us keep the site running.
Note: MacRumors is an affiliate partner with some of these vendors. When you click a link and make a purchase, we may receive a small payment, which helps us keep the site running.
Compared to the Prime Early Access deal, today’s sale is a second-best price on the AirPods Pro 2. They’re in stock today with an estimated delivery window given between November 9 and 12.
Apple Watch Series 8
Additionally, Amazon has the cellular models of the Apple Watch Series 8 for all-time low prices this week. You can get the 41mm cellular Apple Watch Series 8 for $389.00, down from $499.00. This one is available in three colors: (PRODUCT)RED, Midnight, and Starlight aluminum, and each color has both S/M and M/L band sizes available at this price.

The 45mm cellular Apple Watch Series 8 is available for $489.99, down from $529.00. This one is on sale in the same three colors as the 41mm model, including Starlight, Midnight, and (PRODUCT)RED aluminum options.
We’re keeping track of all of the season’s best Apple-related deals in our Black Friday roundup, so be sure to check back throughout the month for an updated list of all the most notable discounts you’ll find for Black Friday 2022.
This article, “Deals: Amazon Has AirPods Pro 2 for $234 ($15 Off) and Cellular Apple Watch Series 8 for $389 ($110 Off)” first appeared on MacRumors.com
Discuss this article in our forums
The cutting-edge cellular therapies aiming to ease America’s organ shortage
Despite being the wealthiest nation on the face of the planet, the United States chronically runs short of transplantable organs. Kidneys are far and away the most sought-after organ for transplantation, followed by livers. While the liver is the only human organ known capable of regenerating itself, if you damage yours badly enough for long enough — as some 30 million Americans have — then the only treatment is a transplant. Assuming you can even acquire one for doctors to stick in you. Every year demand for replacement livers outstrips supply by a scope of tens of thousands.
“Only one-third of those on the liver transplant waiting list will be transplanted, and the demand for livers is projected to increase 23 percent in the next 20 years,” a multidisciplinary team of researchers observed in 2016’s Liver-Regenerative Transplantation: Regrow and Reset. “Exacerbating the organ shortage problem, the donor pool is expected to shrink further because of the obesity epidemic. Liver steatosis [aka fatty liver disease] is increasingly common in donors and is a significant risk factor in liver transplantation.”
To address this critical shortage, the study authors note that doctors have explored a variety of cutting-edge regimens, from cell repopulation and tissue engineering, nanoparticles to genomics, mechanical aids to porcine-derived xenotransplantation, all with varying degrees of success. Cellular repopulation has been used for years, a process that injects healthy liver cells into the patient’s damaged organ through a portal vein where they adhere themselves to the existing cellular scaffolding and grow into new, functional liver tissue.
“Creating an immediately available and inexhaustible supply of functioning liver cells from autologous tissue would allow early intervention in patients with hepatic failure and would allow liver cells to be infused over a longer period of time,” the 2016 study’s authors note. “Combined with recent advances in genome-editing technology, such liver cells could be used widely to treat devastating liver-based inborn errors of metabolism and to eliminate the need for a life-long regimen of immunosuppressive drugs and their complications.” The downside to this technique is the pace at which the donor cells proliferate, making it a poor tool against acute liver failure.
Extracellular Vesicle-based therapies, on the other hand, leverage the body’s intracellular communications pathways to deliver drugs with, “high bioavailability, exceptional biocompatibility, and low immunogenicity,” according to 2020’s Extracellular Vesicle-Based Therapeutics: Preclinical and Clinical Investigations. “They provide a means for intercellular communication and the transmission of bioactive compounds to targeted tissues, cells, and organs” including “fibroblasts, neuronal cells, macrophages, and even cancer cells.”
EVs are the postal letters that cells send one another. They come in a variety of sizes from 30 to 1000 nm and have exterior membranes studded with multiple adhesive proteins that grant them entry into any number of different types of cells. Exploiting the biological equivalent to a janitor’s key ring, researchers have begun tucking therapeutic nanoparticles into EVs and using them to discreetly inject treatments into the targeted cells. However, these treatments are still in the experimental stages and are most effective against acute liver failure and inborn metabolic diseases rather than end-stage liver failure.
Mechanical aids, the hepatocytic equivalent to a dialysis machine, like the Mayo Spheroid Reservoir Bioartificial Liver (SRBAL, above) are ideal for treating cases of acute liver failure, able to take over the entirety of the patient’s liver function externally and immediately. However, such procedures are both expensive and temporary. The SRBAL can only support a patient for up to two weeks, making it more suitable for keeping someone alive until a donor can be located rather than as a permanent, pacemaker-like solution.
The bioprinting and implantation of replacement livers has also shown promise, though they too are still in early development and largely not near ready for widespread adoption. Interspecies transplantation using genetically-engineered pig organs are a bit closer to clinical use, with surgeons successfully transplanting a porcine heart into a human patient for the first time this past January (though he died of complications two months later). Pig kidneys and livers have similarly been transplanted into human recipients, often with less drastic side effects than death.
No matter where the transplanted organ comes from, getting it into the patient is invariably going to involve a significant surgical procedure. However, the Lygenesis company recently unveiled its non-invasive solution: tricking the patient’s body into growing a series of miniature, ectopic liver “organoids” in its own lymphatic system like a crop of blood-scrubbing potatoes.
For those of you who dozed through high school bio, a quick recap of terms. The lymphatic system is a part of the immune system that serves to circulate some 20 liters of lymph throughout your body, absorb excess interstitial fluids back into the bloodstream, and incubate critical lymphocytes like T-cells. Organoids, on the other hand, are biological masses artificially grown from stem cells that perform the same functions as natural organs, but do so ectopically, in that they function in a different part of the body as a regular liver. Blood-scrubbing potatoes are self-explanatory.
“Fundamentally, Lygenesis uses the lymph node, your body’s natural bio reactors typically used for T-cells,” company CEO and co-founder Michael Hufford, told Engadget. “We hijacked that same biology, we engraft our therapies into the lymph nodes to grow functioning ectopic organs.”
“We use an outpatient endoscopic ultrasound procedure where we’re going down through the mouth of the patient using standard endoscopic equipment,” Hufford continued. “We engraft ourselves there in minutes under light sedation, so it’s very low medical risk and also is really quite inexpensive.” He notes that the average cost for a proper, in-hospital liver transplant will set you back around a million dollars. Lygenesis’ outpatient procedure “is billed at a couple of thousand or so,” he said.
More importantly, the Lygenesis technique doesn’t require a full donated liver, or even a large fraction of one. In fact, each donated organ can be split among several dozen recipients. “Using our technology a single donated liver can reach 75 or more patients,” Hofford said. The process of converting a single donated liver into all those engraftable samples takes a team of three technicians more than six hours and 70 steps to complete. The process does not involve any gene manipulation, such as CRISPR editing.
This process is quite necessary as patients cannot donate culturable liver cells to themselves. “Once you have end-stage liver disease, you typically have a very fibrotic liver,” Hofford noted. “It will bleed at the slightest sort of intervention.” Even the simple act of collecting cellular samples can quickly turn deadly if the wrong bit of organ is bisected.
And it’s not only the transplant recipients themselves who are unable to donate. Hofford estimates between 30 and 40 percent of donated livers are too worn to be successfully transplanted. “One of the benefits of our technology is we’re using organs that have been donated but will otherwise be discarded,” he said.
Once engrafted into a lymph node, the liver organoid will grow and vascularize over the course of two to three months, until it is large enough to begin supporting the existing liver. Hufford points out that even with end-stage disease, a liver can retain up to 30 percent of its original functionality, so these organoids are designed to augment and support the existing organ rather than replace it outright.
Lygenesis is currently in Phase 2A of the FDA approval process, meaning that a small group of four patients have each received a single engraftment in a lymph node located in their central body cavity near the liver itself (the body has more than 500 lymph nodes and apparently this treatment can technically target any of them). Should this initial test prove successful subsequent study groups will receive increasing numbers of engraftment, up to a half dozen, to help the company and federal regulators figure out the optimal number of organoids to treat the disease.
While the liver’s inherent regenerative capabilities make it an ideal candidate for this procedure, the company is also developing similar treatments for the kidneys, pancreas and thymus gland as well as inborn metabolic liver ailments like maple syrup urine disease. These efforts are all at much earlier points in development than the company’s end stage liver work. “Within the next five years, we would love to see our liver program submitted to the FDA as a new biologic therapy and be commercially available,” Hufford said. “I think that’d be a realistic timeframe.”
